Introduction
Chemical Constituents
Gypenosides
Polysaccharides, flavonoids, sterols, amino acids, and inorganic elements
Pharmacological Properties
Antineoplastic effects
Anti-atherosclerotic effects
Lipid-modulating effects
Neuroprotective effects
Hepatoprotective effects
Effects on hypoglycemia
Others
Conclusions
Introduction
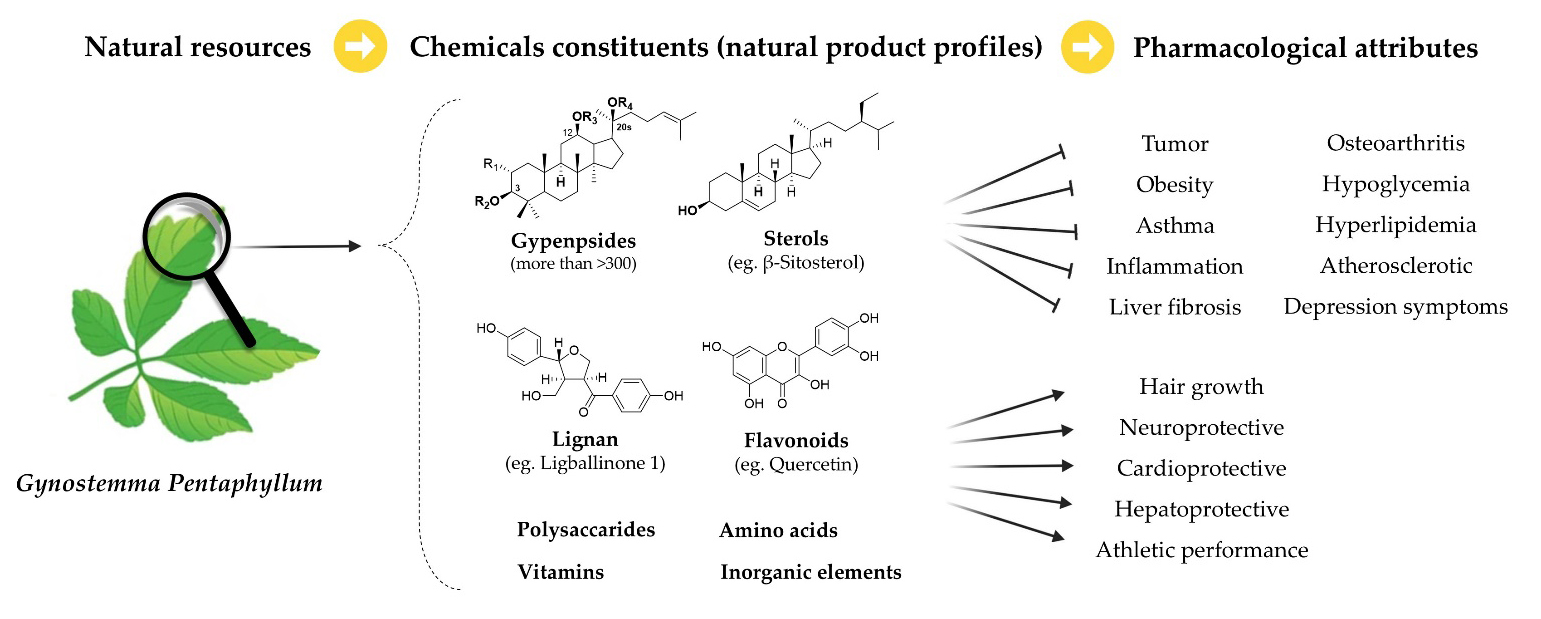
Gynostemma pentaphyllum (Thunb.) Makino is a herbaceous vine in the gourd family (Table 1) that is widely distributed in South and East Asia, and in South Korea, China, and Japan has long been used as a medicinal herb or in preparation of tea. In South Korea, particularly on Jeju Island, the leaves have been utilized for culinary purposes since ancient times, and there is documented evidence of the root of this plant being employed as a medicinal agent for the treatment of hematuria, edema, sore throat, and tumors (Lee et al. 2019). The leaves and aboveground parts of G. pentaphyllum contain high concentrations of dammarane-type saponins, the structural similarity of which to those in ginseng has been well documented (Jang et al. 2001). Whereas the saponins in ginseng are referred to as ginsenosides, those in G. pentaphyllum are classed as gypenosides. G. pentaphyllum also contains some of the ginsenosides found in ginseng, which has led to the plant being referred to as “the poor’s ginseng”, due to its ability to thrive on Jeju Island. To date, more than 316 gypenosides have been isolated, and pharmacologists are continuously discovering gypenosides that had hitherto been unknown to the scientific community. This novel class of saponins have been scientifically assessed for their potential pharmaceutical benefits with respect to inflammation, diabetes, immune function, stress management, asthma, and depression. In this review, we accordingly focus on describing the molecular mechanisms and therapeutic applications of these enigmatic molecules (Chen et al. 2023) based on a comprehensive survey of the literature that has assessed the pharmacological properties of its marker compounds since 2022, identifying the biochemical basis of its biosynthesized chemical constituents and their therapeutic efficacy.
Chemical Constituents
Gypenosides
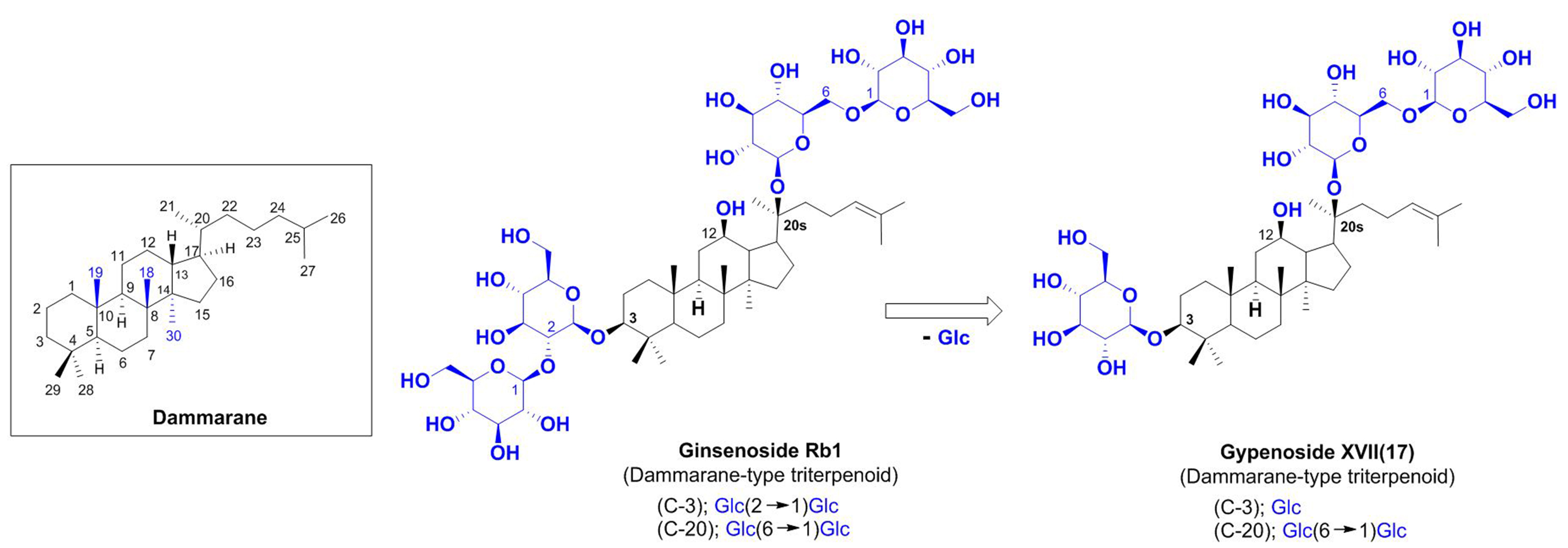
Gypenosides are considered to be the primary bioactive compounds derived from G. pentaphyllum. Molecules in this family have been shown to have anti-cancer, antioxidant, anti-inflammatory, and metabolic modulating properties, mediated via a diverse range of pharmacological mechanisms. Gypenosides have been established to be the active constituents of G. pentaphyllum responsible for the physiological effects observed when using this plant and have been demonstrated to have notable effects in the prevention and treatment of different diseases, particularly cancer, cardiovascular diseases, metabolic disorders, and neurodegenerative diseases (Liu et al. 2021). The structures of all gypenosides are not delineated in this paper; however, they demonstrate a significant structural similarity to ginsenosides, which are the bioactive components of ginseng. Both gypenosides and ginsenosides are named according to their original resources of isolation, yet they differ primarily in the position and type of their sugar moieties. Notably, GypenosideXVII is distinguished by the absence of one glucose unit compared to its ginsenoside counterpart, implying that both may undergo metabolism to yield similar derivatives, specifically compound K and protopanaxadiol (Fig. 1). These compounds are classified as dammarane-type triterpenoids, characterized by a dammarane aglycone core comprising 30 carbons, with hydroxyl groups located at the 3, 12, and 20 positions, and various subtypes arising from diverse glycosidic linkages. This structural homology substantiates their pharmacological potential, which has led to the employment of Jeju's indigenous stonewort as a substitute for red ginseng in traditional medicine.
Polysaccharides, flavonoids, sterols, amino acids, and inorganic elements
In addition to gypenosides, G. pentaphyllum has been established to contain significant quantities of polysaccharides, flavonoids, sterols, amino acids, and inorganic elements. Polysaccharides have been identified as significant bioactive compounds in G. pentaphyllum, with notable immunomodulatory, antineoplastic, antioxidant, and anti-inflammatory properties, particularly with respect to enhancing immune system function, promoting antineoplastic activity, and scavenging free radicals. Flavonoids are a class of plant compounds that have well established antioxidant, anti-inflammatory, cardiovascular protective, and antineoplastic properties, including quercetin and kaempferol, which have been identified as contributing directly to the pharmacological activity of this plant. Sterols serve as the fundamental components of cell membranes, and the phytosterols present in G. pentaphyllum have been demonstrated to play key roles in cholesterol metabolism and cardiovascular health, contributing to reductions in the levels of cholesterol in blood, a factor that is conducive to the maintenance of cardiovascular health. Gynostemma pentaphyllum has also been identified as a rich source of both essential and non-essential amino acids. which are vital for a range of physiological processes within the body, including muscle development, immune system function, neurotransmission, and metabolic processes. Furthermore, G. pentaphyllum contains a range of minerals, including calcium, magnesium, iron, and potassium, which are indispensable for numerous physiological functions in the human body, includeing bone health, muscle function, blood pressure regulation, and redox reactions (Li et al. 2016).
Pharmacological Properties
Antineoplastic effects
The pharmacological effects of G. pentaphyllum have been demonstrated to encompass antineoplastic properties, as evidenced by the extant literature listed in Table 2.
Table 2.
Anti-cancer potential of G. pentaphyllum
| Active ingredient | Models | Mechanism | Ref. |
| Gypenosides | GC-27 and SGC-7901 cells | Induced the apoptosis of gastric cancer cells by inhibiting the PI3K/AKT/mTOR pathway. | 6 |
| Primary mouse CD8+ T cells | Simultaneously inhibited the expression of PD-L1 in gastric cancer cells, thus enhancing the antitumor immunity of T cells. | ||
| Quercetin and rhamnazin | Mouse model of NSCLC | May exert therapeutic effects on NSCLC through the modulation of multiple targets, such as MYC, ESR1, and HIF1A. | 7 |
| Gypenosides | Two cell lines (Huh-7 and Hep3B) | Gyp suppressed mevalonate (MVA) pathway mediated cholesterol synthesis by inhibiting HMGCS1 transcription factor SREBP2. | 8 |
| Rhamnazin, Quercetin | Network pharmacology, molecular docking, and molecular dynamics simulations | By acting on CCND1, RELA, HIF1A, JUN, TNF, and other targets, GP regulates several signaling pathways, including PI3K/Akt, MAPK, cell aging, and TNF signaling pathway. | 9 |
Apoptosis-inducing effects
Gypenosides have been reported to have antineoplastic effects based on their capacity to promote the proliferation, migration, and apoptosis of different cancer cell types. To date, however, there have been no studies conducted to evaluate their efficacy in the treatment of gastric cancer. Recently, however, it has been reported that gypenosides induce the apoptosis of gastric cancer cells by inhibiting the PI3K/AKT/mTOR pathway and simultaneously inhibit the expression of PD-L1 in these cells to enhance the antitumor immunity of T cells, thereby identifying G. pentaphyllum as a source of compounds for a novel immunotherapeutic treatment of gastric cancer (Wu et al. 2024).
Prevention and treatment of non-small-cell lung cancer
Although the anticancer potential of Gynostemma pentaphyllum is not unexpected, the underlying pharmacological mechanisms in the treatment of non-small-cell lung cancer (NSCLC) have yet to be sufficiently determined. In 2022, it was revealed that the therapeutic effects of G. pentaphyllum on NSCLC are mediated via responses to ketones, gland development, and cellular responses to foreign stimuli, with quercetin and rhamnazin being identified as the active constituents that bind stably to specific targets (MYC, ESR1, and HIF1A), as indicated by the findings of molecular docking analyses (Liang et al. 2022).
Inhibition of liver cancer cells
In 2023, Smith et al examined the pharmacological effects of gypenosides identifining a number of notable properties, including antineoplastic, anti-obesity, and hepatoprotective effects. These compounds were demonstrated to limit cholesterol production and target HMGCS1, a protein involved in cholesterol synthesis, the downregulation of which inhibits the progression of hepatocellular carcinoma (HCC) and abnormal cholesterol metabolism. Furthermore, elevated expression of HMGCS1 in human models of HCC has been established to be associated with unfavorable clinical outcomes. The findings of this study accordingly indicated that gypenosides might have potential utility as effective cholesterol-lowering agents, both in the prevention and treatment of HCC, and highlight the significance of targeting the SREBP2–HMGCS1 axis within the MVA pathway as a promising HCC treatment strategy (Xiao et al. 2023).
Therapeutic role in osteosarcoma
Osteosarcoma, a type of malignant bone tumor, poses a serious threat to the health and life of both children and adolescents. Although G. pentaphyllum has been shown to have antitumor properties in this regard, the precise mechanisms underlying these effects remain to be fully elucidated. Recently (2024), however, network pharmacology, molecular docking, and molecular dynamics simulation approaches have been adopted with a view toward clarifying the molecular mechanisms of the marker compounds of G. pentaphyllum in the treatment of osteosarcoma, identifying quercetin and rhamnazin as the two most important constituents in this respect. Moreover, it has been demonstrated that the observed therapeutic effects are mediated via a promotion of apoptosis, cell cycle regulation, and control of the cellular responses to tumor necrosis factor (Zhang et al. 2024).
Anti-atherosclerotic effects
The cardiovascular protective effects of Gynostemma pentaphyllum have garnered particular scientific interest in recent years, as evidenced by the summary of recent studies presented in Table 3. For example. gypenoside XLIX has been demonstrated to mitigate atherosclerosis in a model of high-fat choline diet (HFCD)-induced atherosclerosis in ApoE−/− mice, a phenomenon attributable to modulation of the gut microbiota. The findings of this study, indicating that modulation of the gut microbiome may play pivotal role, are considered to be of particular importance given their potential implications for the prevention and treatment of cardiovascular disease (Gao et al. 2022).
Table 3.
Anti-Atherogenic Effect of G pentaphyllum
| Active ingredient | Models | Mechanism | Ref. |
| Gypenoside XLIX | High-fat choline diet (HFCD)-induced atherosclerosis model in ApoE−/− mice | Decrease Verrucomicrobia, Proteobacteria, and Actinobacteria abundance, and increase Firmicutes and Bacteroidetes population. Moreover, the Firmicutes/Bacteroidetes ratio increased significantly after treatment with GPE. After treatment with GPE, the relative abundance of trimethylamine-producing intestinal bacteria Clostridioides and Desulfovibrionaceae decreased while butyrate-producing bacteria such as Eubacterium, Roseburia, Bifidobacterium, Lactobacillus, and Prevotella increased significantly. | 10 |
Lipid-modulating effects
The findings of recent studies have provided evidence for the efficacy of gypenosides, in the prevention and treatment of metabolic diseases, the supporting literature for which is listed in in Tables 4 and 5.
Table 4.
Effect on Hyperlipidemia of G. pentaphyllum
| Medicinal composition | Models | Mechanism | Ref. |
| G. pentaphyllum saponins | Molecular docking and cell experiments | G. pentaphyllum improves hyperlipidemia by mediating the LOX1-PI3K-AKT-eNOS pathway. | 11 |
| Gypenosides (Gyps) in combination with capsaicinoids (Caps) | High-fat-diet (HFD)-induced rat | Gyps/Caps combination lowered the liver weight, liver index, serum lipid profile and hepatic cholesterol content, improved liver function by reducing the serum markers of alanine aminotransferase (ALT) and aspartate aminotransferase (AST), thereby relieving the hepatic injury. Moreover, Gyps/Caps combination inhibited oxidative stress by decreasing the lactic dehydrogenase (LDH) activity and increasing the superoxide dismutase (SOD) activity. | 12 |
| Gypenoside aglycone (Agl) | Molecular docking and cell experiments | G. pentaphyllum improves hyperlipidemia by mediating the LOX1-PI3K-AKT-eNOS pathway. | 13 |
Table 5.
Anti-Obesity Effect of G. pentaphyllum
| Medicinal composition | Models | Mechanism | Ref. |
| G. pentaphyllum gold nanoparticles (GP-AuNPs) | 3T3-L1 obese, Raw 264.7 macrophage cells | Significantly decreased lipid accumulation in 3T3-L1 obese and reduced NO production in Raw 264.7 macrophage cells. Decreased the face of these genes remarkably, revealing the antiadipogenic and anti-inflammatory activity of our synthesized GP-AuNPs. | 14 |
| Gypenoside IX | Hypertensive rats | Gypenoside IX inhibits the renin–angiotensin system activation and oxidative stress through the inactivation of the mitogen-activated protein kinase pathway in obesity-mediated hypertension in rats. | 15 |
| Gypenoside XXVIII_qt | Hypertensive rats | Acts on obesity through multiple targets and pathways, with TP53, AKT1, TNF and JUN identified as key core targets. | 16 |
Effects on hyperlipidemia
Until recently, the therapeutic efficacy and the mechanism of action of G. pentaphyllum in the treatment of hyperlipidemia had remained insufficiently established. However, in 2024, cellular experiments revealed that extracts of this plant mitigate hyperlipidemia by modulating the expression of pivotal proteins in the LOX1–PI3K–AKT–eNOS pathway (Shen et al. 2024). Previously, in 2023, it had been reported that the combination of gypenosides and capsaicinoids contributed to an amelioration of fatty liver and hyperlipidemia via an enhancement of liver function and suppression of antioxidant stress in a hyperlipidemic model (Zhang et al. 2023), and a team of researchers from China have demonstrated that gypenoside aglycones (Agl) have the capacity to cause reductions in the levels total cholesterol, triglycerides, and low-density lipoprotein cholesterol, whilst concurrently ameliorating the effects of high-fat diet-induced liver damage. The molecular analyses performed by these authors revealed the underlying regulatory mechanisms involving the regulation of key lipid metabolism genes and proteins by Agl, thereby indicating the potential clinical application of Agl in the management of hyperlipidemia (Xie et al. 2024).
Anti-obesity effects
The first study to investigate the efficacy of G. pentaphyllum gold nanoparticles (GP-AuNPs) against obesity and related inflammation was conducted in 2022 in South Korea. In vitro assays of these nanoparticles revealed a significant reduction in lipid accumulation in 3T3-L1 mice and a reduction in NO production in RAW 264.7 macrophages. Moreover, significant reductions in the expression of two important adipogenic genes (PPARγ and CEPBα) and a major inflammatory cytokine (TNF-α) provided evidence to indicate the anti-adipogenic and anti-inflammatory activities of the synthesized GP-AuNPs (Akter et al. 2022).
Using a rat model of hypertension, the effects and mechanisms of gypenoside IX on obesity-related hypertension have also been examined, revealing that the administration of gypenoside IX contributed to alleviating this condition, as evidenced by reductions in both systolic and diastolic blood pressure, as well as a reduction in body weight. Furthermore, gypenosides derived from G. pentaphyllum were found to alleviate pathological symptoms in the myocardium and aorta. In the context of obesity-mediated hypertension in rats, inactivation of the mitogen-activated protein kinase pathway has been observed to impede renin-angiotensin system activation and oxidative stress (Meng et al. 2022). It has also been demonstrated that gypenoside XXVIII_qt exerts its effects on obesity by modulating multiple target pathways, resulting in alterations in the structure of the gut microbiome and augmentation of the abundance and diversity of the mucosal microbiome, consequently resulting in a reduction in weight gain in obese mice (XIE and DENG 2024).
Neuroprotective effects
The neuroprotective effects of G. pentaphyllum have been particularly noted with respect to neurodegenerative brain diseases such as Alzheimer's and Parkinson's diseases, as outlined in Table 6, with neuroprotective properties of the gypenosides derived from this plant being shown to be mediated via a modulation of key signaling pathways, such as the NF-κB, Nrf2, and AKT pathways (Liang et al. 2024).
Table 6.
Neuroprotective Effect of G. pentaphyllum
| Active ingredient | Mechanism | Ref. |
| Gypenoside | Pharmacological studies have discovered that gypenosides can modulate various major signaling pathways like NF-κB, Nrf2, AKT, ERK1/2, contributing to the neuroprotective properties. | 17 |
Hepatoprotective effects
The hepatoprotective effects of Gynostemma pentaphyllum have also been studied using different disease models, as shown in Table 7.
Table 7.
Hepatoprotective Effects of G. pentaphyllum
| Active ingredient | Models | Mechanism | Ref. |
| Gypenoside XLVI | Chronic liver injury mice | XLVI can inhibit TGF-β-induced activation of hepatic stellate cells and ECM deposition in vitro. The underlying mechanism study verified that it upregulated the protein expression of protein phosphatase 2C alpha and strengthened the vitality of the phosphatase together with a PP2Cα agonist gypenoside NPLC0393. | 18 |
| Gypenoside A (GPA) | Experimental carbon tetrachloride (CCl4)-induced liver disease model | Involving the upregulation of the PDK1/Bcl-2 signaling pathway by GPA, thereby preventing cell apoptosis. | 19 |
Liver fibrosis
Liver fibrosis, caused by chronic liver damage, has emerged as one of the pre-eminent global health concerns. Among the active saponin constituents isolated from G. pentaphyllum that have been found to have hepatoprotective effects is gypenoside XLVI, which was demonstrated to inhibit the accumulation of extracellular matrix in hepatocytes in a chronic liver injury model and to alleviate liver fibrosis by inhibiting TGF-β-induced hepatocyte activation (Li et al. 2023).
CCl4-induced liver injury
In a CCl4-induced liver injury model, gypenosides derived from G. pentaphyllum have been demonstrated the have the capacity to impede apoptosis via the PDK1/Bcl-2 pathway. Moreover, in 2024, a research team conducted a comparative analysis of the effects of gypenoside A from G pentaphyllum and ginsenoside Rb3 from ginseng in the same experimental setting, which revealed that the former compound had superior efficacy with respect to reducing hepatocyte death, thereby indicating that G pentaphyllum may have a more pronounced hepatoprotective effect than ginseng (Hu et al. 2024).
Effects on hypoglycemia
Diabetes, a chronic condition characterized by abnormally high levels of glucose in the blood, arises from a deficiency or impaired functionality of the hormone insulin. Left untreated, diabetes can lead to serious complications, thus highlighting the need for hypoglycemic drugs such as metformin and sulfonylureas. However, these pharmacotherapies are associated with significant adverse events, and, consequently, there is growing interest in the development of new diabetic drugs derived from natural products that have long-lasting effects and fewer side effects. In 2024, G. pentaphyllum was proven to have significant biological activity in the prevention and treatment of type 2 diabetes mellitus (T2DM), with the underlying mechanism of action associated with an improvement in insulin resistance being demonstrated to be mediated by an inhibition of gluconeogenesis via insulin resistance-related PI3K/Akt and FoxO1 signaling pathways, which are closely associated with T2DM (Table 8). Accordingly, the imminent development of a G. pentaphyllum-based strategy for the treatment of diabetes is widely anticipated (Yang et al. 2024).
Table 8.
Hypoglycemic Effect of G. pentaphyllum
| Active ingredient | Models | Mechanism | Ref. |
|
32 Active ingredients and potential targets of G. pentaphyllum | HepG2 cells | Improve insulin resistance by promoting glucose uptake and glycogen synthesis and inhibiting gluconeogenesis through regulating the IRS1/PI3K/Akt signaling pathway. | 20 |
Others
In addition to the aforementioned biological activities, G. pentaphyllum has been established to have a diverse range of other benefits, including the alleviation of depression-like symptoms, anti-inflammatory effects, management of asthma, enhancement of athletic performance, stimulation of hair growth, cardioprotective benefits, and the treatment of osteoarthritis, a comprehensive summary of which is presented in Table 9.
Table 9.
Others bioactivities of G. pentaphyllum
| Active ingredient | Models | Mechanism | Ref. |
| Gypenoside XVII | C3 in mice exposed to chronic unpredictable mild stress (CUMS) | Significantly attenuated depression-like behaviors in the forced swimming test, tail suspension test and sucrose preference test. also alleviated the acute stress-induced hyperactivity of serum corticosterone levels. significantly inhibited the activation of microglia and the expression of C3 in mice exposed to chronic unpredictable mild stress (CUMS). | 21 |
|
Gypenoside XVII (Gyp XVII) | lipopolysaccharide (LPS)-induced murine RAW 264.7 macrophages and the xylene-induced acute inflammation model of mouse ear edema | Improved anti-inflammatory activity, which significantly inhibited the generation of TNF-α and IL-6 more effectively than its precursor ginsenoside Rb1. | 22 |
| Gypenoside A | BALB/c mice by ovalbumin injection | Gypenoside A treatment can suppress eosinophil infiltration in the lungs, reduce tracheal goblet cell hyperplasia, and attenuate AHR. Gypenoside A significantly reduced Th2 cytokine expression and also inhibited the expression of inflammatory genes and proteins in the lung and bronchoalveolar lavage fluid. In addition, gypenoside A also significantly inhibited the secretion of inflammatory cytokines and chemokines and reduced oxidative expression in inflammatory tracheal epithelial cells. | 23 |
| G. pentaphyllum dried leaf extract equivalent | Healthy untrained young males | Improved mitochondrial respiration, altered AMPK and ACC, and decreased plasma leptin and glucose levels. | 24 |
| G. pentaphyllum extract (GPE) Gypenoside L (GL) | ExGPE (exercise with 300 mg/kg body weight/day of GPE) mice | GPE improved the exercise endurance and capacity in treadmill-trained mice, increased glucose and triglycerides, and decreased the serum creatine kinase and lactate levels after intensive exercise. | 25 |
| Damulin B | Mice | GPHD and damulin B contribute to the hair growth-inducing properties of dermal papilla cells through the AKT/β-catenin signaling pathway. | 26 |
| Gypenosides (GYPs) | Dox-stimulated H9c2 rat cardiomyocytes | Phosphorylation of PI3K, Akt, GSK-3β, and the protein level of Mcl-1 was upregulated by GYP treatment. | 27 |
| Gypenosides | OA Rat Model | GP inhibited key inflammatory mediators, including interleukin (IL)-1β, IL-6, matrix metalloproteinases (MMP)-3 and MMP-13, cyclooxygenase-2, and prostaglandin E receptor 2, surpassing the effects of active controls. | 28 |
Alleviation of depression-like symptoms
The active constituent gypenoside XVII isolated from G. pentaphyllum has been shown to attenuate lipopolysaccharide-induced inflammation in BV2 microglia. Furthermore, assessment of the in vivo effects of gypenoside XVII in alleviating depression-like symptoms and the potential involvement of complement regulation in the antidepressant-like effects revealed significant improvements in depression-like symptoms and production of antidepressant-like effects in mice by inhibiting complement C3/C3aR/STAT3/cytokine signaling in the prefrontal cortex (Zhang et al. 2022).
Anti-inflammatory effects
A comparison between gypenoside XVII and its precursor ginsenoside Rb1 revealed the former compound to have enhanced anti-inflammatory activity, with a significantly more effective inhibition of TNF-α and IL-6 production compared with its precursor, thereby indicating that gypenoside XVII may have superior anti-inflammatory properties compared with ginsenoside Rb1. Enzymatic modification provides a novel approach to the production of more active pharmaceutical constituents and offers the potential for large-scale production of gypenoside XVII as a potent new anti-inflammatory drug candidate (Zhou et al. 2023).
Effects on asthma
Asthma, a chronic respiratory condition, is characterized by inflammation of the bronchial tubes, leading to respiratory distress, in which the bronchial tubes become hypersensitive and constricted in response to external stimuli, or due to an excessive production of mucus. Experimental findings in this regard have revealed that gypenoside A can effectively mitigate airway inflammation and hyper-responsiveness in asthmatic individuals, which can be attributed to an attenuation of inflammatory and oxidative responses in inflamed tracheal epithelial cells and a diminishing of Th2 cell activation (Huang et al. 2022).
Enhancement of athletic performance
The effects of natural compounds on athletic performance represent a prominent area of interest in both the sports and research communities. In 2023, Australian researchers obtained evidence to indicate the efficacy of G. pentaphyllum in enhancing physical metrics associated with athletic performance. Specifically, healthy young males who were given a dried G. pentaphyllum leaf extract reported enhancements in exercise performance, which were established to be associated with enhanced mitochondrial respiration, modulation of the AMPK and ACC pathways, and reduced plasma levels of leptin and glucose (Nayyar et al. 2023). Furthermore, researchers in South Korea have found that rats treated with an extract of G. pentaphyllum were characterized by a significant reduction in fat mass percentage and an increase in lean muscle mass compared with the control rats. These findings thus indicate that G. pentaphyllum may enhance exercise performance and have anti-fatigue effects (Kim et al. 2022).
Stimulation of hair growth
Alopecia areata is a prevalent condition, the pharmacotherapeutic treatments of which tends to be characterized by adverse events. In 2024, researchers in South Korea demonstrated the stimulation of hair growth, both in vitro and in vivo, in response to the administration of G. pentaphyllum leaf hydrodistillate (GPHD). These workers established that the principal active constituent of GPHD is damulin B, and it is this molecule that contributes to promoting hair growth in vitro by stimulating dermal papillary cells via the AKT/β-catenin signaling pathway (Kovale et al. 2024).
Cardioprotective effects
It has also been demonstrated that gypenosides have a certain efficacy in preserving mitochondrial homeostasis and mitigating the development of heart failure. The potential targets of gypenosides and heart failure treatment have previously been examined using a network pharmacology approach, followed by drug-disease target prediction and enrichment analyses. The findings accordingly indicated that gypenosides are characterized by cardioprotective effects mediated via the rescue of defective mitophagy, thereby providing evidence to indicate that PI3K/Akt/GSK-3β/Mcl-1 signaling plays an important role in this process (Zheng et al. 2024).
Effects on osteoarthritis
Osteoarthritis is a condition involving inflammation, pain, joint cartilage damage, synovitis, and irreversible disability. In 2024, using a rat model of induced osteoarthritis, a team of researchers from South Korea confirmed the pain relief, bone function improvement, and chondroprotective effects conferred by G. pentaphyllum. It is anticipated that these results will serve as the foundation for the discovery of pharmaceutical candidates for the effective treatment of osteoarthritis (Jo et al. 2024).
Conclusions
Gynostemma pentaphyllum, renowned for its multifaceted therapeutic properties such as antihypertensive, antioxidant, anti-inflammatory, and anticancer effects, has been the focal point of extensive scientific inquiry. Recently, its research scope has broadened to explore potential benefits in alleviating depressive symptoms, enhancing athletic performance, managing osteoarthritis, and mitigating asthma symptoms. Despite its promise, the path to clinical application presents challenges. The pharmacokinetic constraints of gypenosides, its active constituents, necessitate further investigation to refine their drug-likeness. Critical studies on safety, dosage optimization, and bioavailability are imperative to develop standardized, efficacious formulations. Innovations in drug delivery and bioengineering hold the potential to elevate compound bioavailability, while environmental variability remains a hurdle to consistency and efficacy. Addressing these complexities is crucial to unlocking the full clinical potential of G. pentaphyllum. With the advent of new technologies unveiling unexplored biological mechanisms and pharmacodynamics, the horizon for developing clinical drugs and formulations harnessing G. pentaphyllum appears promising. Fig. 2 encapsulates the essential findings and implications of this study.